Adiabatic Invariants
Physical laws--the basic ones and those derived from them--usually make precise, quantitative statements, like F=ma, E=mc2, E= hν. Very rarely does one encounter a law by which something is approximately true but not exactly (even though the approximation may be very good), e.g. valid for a long time but not to all eternity. Adiabatic invariance is of this kind.
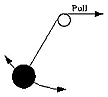
Imagine a pendulum formed by a weight tied to a string, swinging back and forth under the influence of gravity. In each oscillation, as it descends to the bottom of its swing, it gains kinetic energy, then after having passed bottom, it slows down again, to stop briefly at the top of each swing. In the absence of friction and air resistance, one expects its total energy E (kinetic + potential) to stay unchanged, and the frequency ν and period T=1/ν of the oscillation will stay constant, too.
If the string starts at a pulley (see drawing) and is slowly pulled up, the length of the pendulum will gradually decrease, and since a shorter pendulum has a higher frequency ν and a shorter period T, these quantities are also expected to change. But the energy E grows too, because extra work is being done against the force stretching the string, so the swing becomes more vigorous. Suppose the string is drawn up until its length is reduced by a certain fraction--say, by half. It can be shown that the change in the ratio E/ν can be made as small as we wish by making the drawing-up process slow enough.
The ratio is not a real constant of the motion or "invariant" (like the energy in the undisturbed pendulum), but an approximate one, an "adiabatic invariant". The lack of constancy may be attributed to the fact that the tension in the string in any swing does not vary symmetrically--a small asymmetry is added by the fact the string is constantly being shortened, but slowing down the pull greatly reduces the asymmetry and its accumulated effects.
Notice here that the adiabatic invariant E/ν has the same dimensions of joule-sec (i.e. same combination of measurable quantities--also called "action") as the elusive Planck's constant h.
Adiabatic invariants occur in all sorts of periodic motions, including Kepler motion, which was taken as the model of the motion of an electron around the nucleus. Some physicists, notably Paul Ehrenfest, guessed that perhaps electron orbits were stable when the adiabatic invariant I of their motion equaled h times some whole number. For circular orbits in an atom, only one adiabatic invariant I existed, and allowing it to only take the values I=nh with n=1,2,3... gave orbits with energies which exactly matched Balmer's hydrogen spectrum.
In the end it turned out that the quantization of angular momentum, rather than of I, gave a better qualitative understanding of atoms of more than one electron. That is discussed in the section that follows.
Adiabatic invariants again attracted attention in the 1950s, in the study of the motion of ions and electrons in rarefied plasmas. A plasma is a gas hot enough to contain an appreciable fraction of detached ions and electrons, and if it is rarefied enough--as is, for instance, the plasma in the "magnetosphere" surrounding Earth--these particles may move independently for long times before recombining. In the presence of a magnetic field strong enough, they tend to spiral around magnetic field lines, which is a periodic motion, with its specific adiabatic invariants (in fact, several periodicities and invariants exist). The preservation of those invariants turned out to be essential to the trapping of particles in the magnetic fields of Earth and other planets, and also to many plasma phenomena in space and in the lab. But this application of the invariants was part of Newtonian ("classical") mechanics, not involving Planck's constant h but describing phenomena on a scale much larger than atomic dimensions. For more about this direction of research, see the exposition "Exploration of the Earth's Magnetosphere", to which the files linked above belong.